
Congressman Rudy Yakym (R-IN-02) led a bipartisan letter with Reps. Blake Moore (R-UT-01), Suzan K. DelBene (D-WA-01), Terri Sewell (D-AL-07), Lori Trahan (D-MA-03) and Gus Bilirakis (R-FL-12), urging Department of Health and Human Services Secretary Kennedy and Administrator for the Centers for Medicare & Medicaid Services, Mehmet Oz to take action on improving Medicare coverage for FDA-designated breakthrough medical devices.
The letter was signed by 82 Members of Congress and received support from leading health and industry organizations.
"When the FDA designates a device as a breakthrough technology, that means that there is nothing similar already available to patients having the potential to change or save lives,” Rep. Yakym said. “Seniors on Medicare shouldn't have to wait years to access it simply because of bureaucratic processes. This letter makes clear that Congress expects action, and I will keep pushing until patients in Indiana and across the country can get the care they need without unnecessary delays."
“There is no reason that Medicare patients should have to wait to access breakthrough medical devices already cleared by the FDA. The Ensuring Patient Access to Critical Breakthrough Products Act ensures patients receive the most advanced treatments on the market as soon as possible,” Rep. Moore said. “Easing the pathway for Medicare approval has long been a bipartisan priority, and I look forward to working with Secretary Kennedy to ensure this crosses the finish line for our seniors.”
“Seniors should not be forced to wait years for cutting-edge, safe, and effective treatments that could change or even save their lives,” Rep. DelBene said. “Guaranteeing Medicare coverage of breakthrough medical devices would encourage future innovation and investment in next-generation medical technologies while improving the quality of life for millions of seniors. We can’t let the coverage approval process be a barrier to life-saving treatments or care.”
“All Americans deserve access to life-saving, breakthrough therapies and devices,” Rep. Sewell said. “This letter takes a critical step in the right direction to ensure that the review and approval process for breakthrough therapies does not inhibit patient access to innovative and effective technologies for our seniors.”
"When the FDA designates a device as a breakthrough, it means it has the potential to change or even save a patient's life,” Rep. Trahan said. “Medicare seniors shouldn't have to wait years to access treatments that have already cleared that bar. This bipartisan effort is about making sure our coverage policies keep pace with medical innovation, so patients here in Massachusetts and across the country can get the care they need without unnecessary delays."
This effort has earned broad support from industry leaders and patient advocacy organizations across the healthcare community, including MDMA, AdvaMed, Alzheimer's Association, Roche Diagnostics, National Health Council, HealthyWomen, Alzheimer's Impact Movement, Siemens Healthineers, and Indiana Life Sciences Association.
"MDMA thanks Congressman Yakym and all of the signatories for their ongoing bipartisan work to help remove barriers for patients and providers to health care innovation, and to improve access to the lifesaving and life-changing technologies that they need," Mark Leahey, President and CEO of MDMA said. "We will continue to work with Congress, the administration and the broad coalition committed to enacting policies that will strengthen the United States' global leadership in the development of medical technologies."
"Medicare patients shouldn’t have to wait years to benefit from innovations the FDA has already determined to be safe and effective,” Scott Whitaker, president and CEO, AdvaMed said. “Both Congress and the Administration have an opportunity to fix this longstanding gap, strengthen America’s leadership in medical technology, and ensure Medicare policy keeps pace with modern medicine. We appreciate the considerable progress so far and look forward to continuing to work with Congress and the executive branch on a durable solution. Timely access to medtech can make all the difference for patients."
"Indiana is a global leader in medical technology innovation, and Hoosier companies are developing devices that improve and save lives every day,” Kristin Jones, President and CEO, Indiana Life Sciences Association said. “Patients should not face unnecessary delays accessing FDA-designated breakthrough technologies simply because reimbursement pathways lag behind innovation. We applaud Congressman Rudy Yakym for leading efforts to modernize Medicare coverage and ensure seniors can benefit more quickly from next-generation medical devices."
You can find the full letter HERE.
Background:
This Congress, Rep. Yakym introduced H.R. 5343, the Ensuring Patient Access to Critical Breakthrough Products Act alongside Representatives Blake Moore (R-UT-01), Suzan DelBene (D-WA-01), Terri Sewell (D-AL-07), Gus Bilirakis (R-FL-12), and Lori Trahan (D-MA-03). The bill would create a streamlined pathway for FDA-designated breakthrough devices to receive transitional Medicare coverage while CMS conducts a review for a permanent coverage determination, require CMS to assign payment codes within three months of FDA approval, and direct CMS to create a pathway for continued coverage after the transitional period. The legislation passed the Ways and Means Committee by a bipartisan vote of 38-3.


 Indiana FSSA announces results of HCBS attendant care audits - $200 Million in improper payments
Indiana FSSA announces results of HCBS attendant care audits - $200 Million in improper payments
 Applications being accepted for Governor’s 2026 Century and Half Century Business Awards
Applications being accepted for Governor’s 2026 Century and Half Century Business Awards
 Indiana BMV extending hours for primary election
Indiana BMV extending hours for primary election
 $675 million impact reported from initial Regional Economic Development funding
$675 million impact reported from initial Regional Economic Development funding
 PHMS Amazing Shake
PHMS Amazing Shake
 Parke Heritage students in competition at FFA District Contest
Parke Heritage students in competition at FFA District Contest
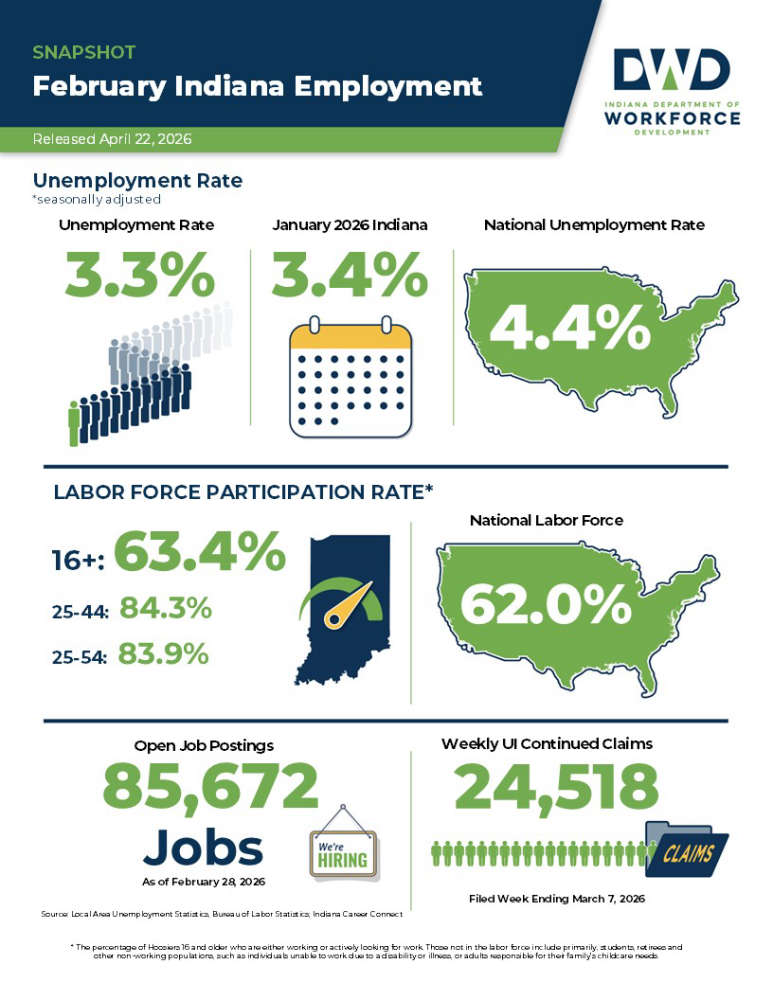 Indiana's February unemployment rate below national average
Indiana's February unemployment rate below national average
 Indiana hiring five county executive directors in training positions
Indiana hiring five county executive directors in training positions
 Be on the lookout for farm equipment on the road
Be on the lookout for farm equipment on the road
 SW Parke projected to save over a million dollars closing Montezuma Elementary
SW Parke projected to save over a million dollars closing Montezuma Elementary
 Changes coming to South Vermillion School Corporation's cell phone policy
Changes coming to South Vermillion School Corporation's cell phone policy
 Crawfordsville man faces list of felony child sex charges
Crawfordsville man faces list of felony child sex charges
 Domestic incident results in arrest in Parke County
Domestic incident results in arrest in Parke County
 Clinton man nabbed in burglary case
Clinton man nabbed in burglary case
 Indiana releases update to the State Nutrient Reduction Strategy
Indiana releases update to the State Nutrient Reduction Strategy
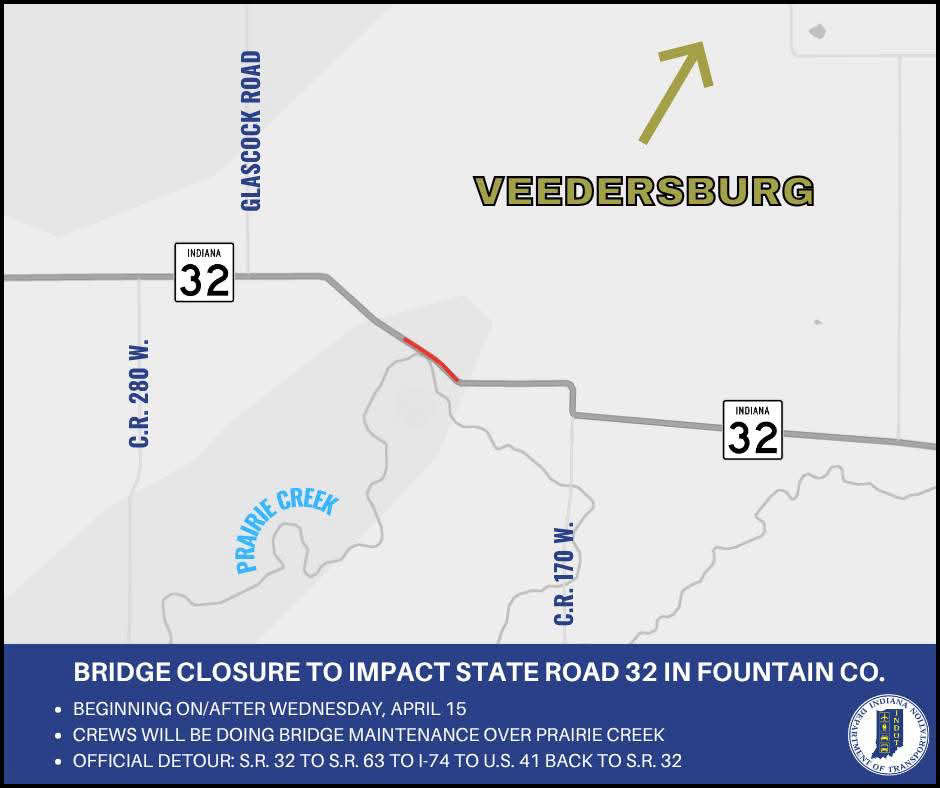 Bridge closure to impact State Road 32 over Prairie Creek in Fountain County
Bridge closure to impact State Road 32 over Prairie Creek in Fountain County




